This simple experiment allows you to investigate yeast respiration.
Materials
These are the materials you need:
- Dry yeast
- Sugar
- Warm water
- Balloon
- Two small plastic bottles or jars
- Rubber band
- Thermometer
Methodology
Now, proceed as follows:
Add one teaspoon of sugar and one teaspoon of dry yeast to a small plastic bottle or jar.
Add about 100 ml of warm water to the bottle and stir until the yeast and sugar are dissolved.
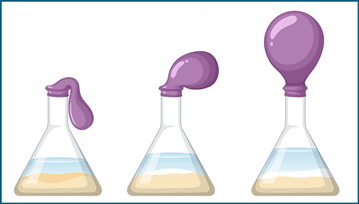
Attach a balloon to the top of the bottle using a rubber band.
Repeat steps 1-3 with a second bottle, but do not add any sugar to this bottle.
Place both bottles in a warm place (around 30-35°C) and wait for 10-15 minutes.
Observe the balloons on both bottles. Which one has inflated more?
Use a thermometer to measure the temperature inside both bottles. Were they both at the same temperature?
Explanation
Yeast can respire in the presence or absence of oxygen. When yeast is added to warm water and sugar, it will begin to respire and release carbon dioxide gas as a waste product. The carbon dioxide gas will inflate the balloon attached to the bottle. The more the yeast respires, the more carbon dioxide is produced and the more the balloon inflates.
In the second bottle, where no sugar was added, the yeast does not have any glucose to respire. Therefore, no carbon dioxide gas is produced and the balloon remains uninflated.
By measuring the temperature inside both bottles, you can find out that the yeast is not affected by the temperature differences, and that the difference in balloon inflation is due to the presence or absence of glucose.

